|
11/11/2023 0 Comments Timi trials 
standard-dose prophylactic anticoagulation and antiplatelet therapy vs. GOLDILOX is a Phase IIB, Randomized, Double-Blinded, Placebo-Controlled Trial To Evaluate the Efficacy and Safety of MEDI6570 (a fully human monoclonal antibody against LOX-1) in Participants with a Prior Myocardial Infarction, Persistent Inflammation, and Elevated N-Terminal Prohormone Brain Natriuretic Peptide.ĪZALEA – A phase 2b, multicenter, randomized, active-controlled study to evaluate the safety and tolerability of two blinded doses of the novel factor XI inhibitor abelacimab compared with open-label rivaroxaban in patients with atrial fibrillation.ĬOVID-PACT – a multicenter, randomized, open-label, 2 x 2 factorial design trial of full-dose anticoagulation vs. VESALIUS-CV – assessing the effect of lowering low-density lipoprotein cholesterol (LDL-C) with evolocumab on major cardiovascular events in adults without a prior myocardial infarction (MI) or stroke who are at high risk of a cardiovascular event.ĭAPA ACT HF – an investigator-initiated, randomized, double-blind, placebo-controlled trial in patients with heart failure who have been stabilized during hospitalization for acute heart failure, evaluating the effect of in-hospital initiation of dapagliflozin on the clinical outcome of cardiovascular death or worsening heart failure. 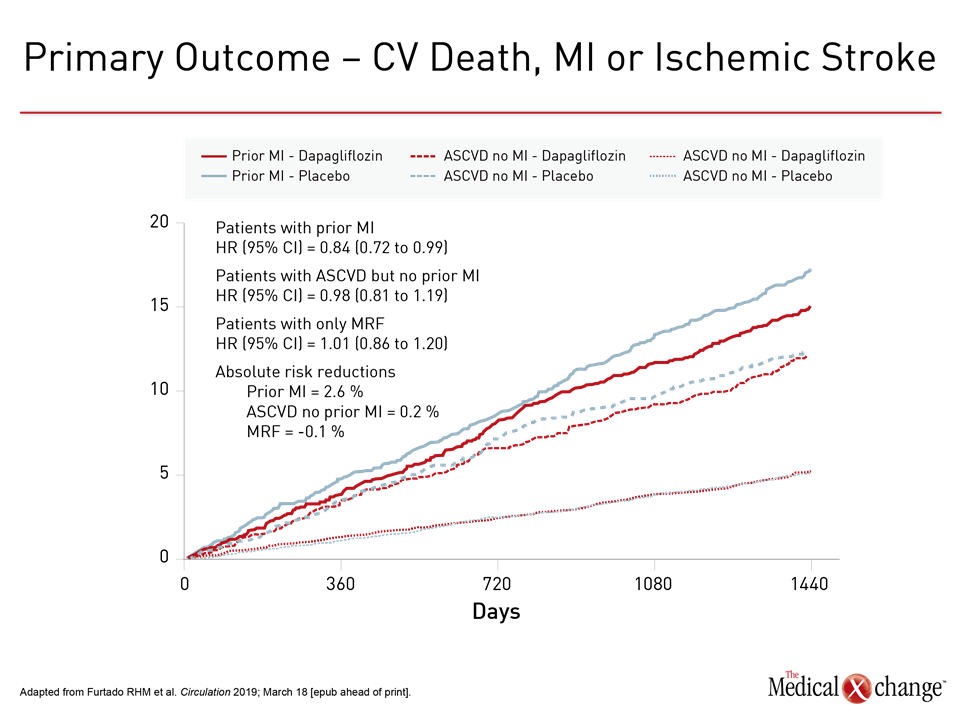
TIMI risk can be calculated on the TIMI website under "Clinical Calculators.ORION 4 – examining the effects of inclisiran, a small interfering RNA (siRNA) that inhibits the synthesis of PCSK9, on cardiovascular outcomes among individuals with atherosclerotic cardiovascular disease. 'TIMI risk' estimates mortality following acute coronary syndromes. 1 Pointĥ% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ĩ% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.ġ3% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ģ0% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ģ6% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization.Ĥ1% risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization. % risk at 14 days of: all-cause mortality, new or recurrent MI, or severe recurrent ischemia requiring urgent revascularization. *Risk factors include: family history of CAD, hypertension, hypercholesterolemia, diabetes, or being a current smoker. Severe angina (≥ 2 episodes w/in 24 hrs).Known coronary artery disease (CAD) (stenosis ≥ 50%).TIMI Score Calculation (1 point for each): In patients with UA/NSTEMI, the TIMI risk score is a prognostication scheme that categorizes a patient's risk of death and ischemic events and provides a basis for therapeutic decision making. Among the group's most important works is the TIMI Risk Score, which assesses the risk of death and ischemic events in patients with unstable angina (UA) or non-ST elevation myocardial infarction (NSTEMI). Braunwald held the chairmanship until 2010, when he appointed Marc Sabatine to the position. The TIMI Study Group was founded by physician Eugene Braunwald in 1984. The group has its headquarters in Boston, Massachusetts. The Thrombolysis In Myocardial Infarction ( TIMI) Study Group, is an academic research organization (ARO) affiliated with Brigham and Women's Hospital and Harvard Medical School with a focus in the field of cardiovascular disease. For other uses, see Timi (disambiguation). For the IBM technology, see IBM i § TIMI. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
